Compliance in healthcare marketing is one of those topics that makes people groan. It feels like a wall between you and every clever campaign idea you've had. But here's the thing, that wall isn't as solid as you think. Most independent pharmacies and clinics that struggle to grow digitally aren't held back by regulations, they're held back by misunderstanding them. This guide cuts through the confusion, lays out exactly what the rules require, and shows you how to run marketing that builds patient trust, avoids legal exposure, and actually drives growth.
Table of Contents
- Why compliance is critical in healthcare marketing
- Key regulations shaping healthcare marketing
- Common compliance pitfalls and how to avoid them
- How compliance enables smarter marketing and growth
- Why the best healthcare marketers see compliance as an asset, not a restraint
- Get hands-on help with compliant healthcare marketing
- Frequently asked questions
Key Takeaways
| Point | Details |
|---|---|
| Compliance builds trust | Following advertising laws enhances patient confidence and loyalty. |
| Know your regulations | Multiple federal and state rules impact every healthcare marketing effort. |
| Avoid costly pitfalls | Simple missteps can lead to fines, so regular compliance checks are critical. |
| Leverage compliance for growth | A systematic compliance approach unlocks safer, smarter patient engagement. |
Why compliance is critical in healthcare marketing
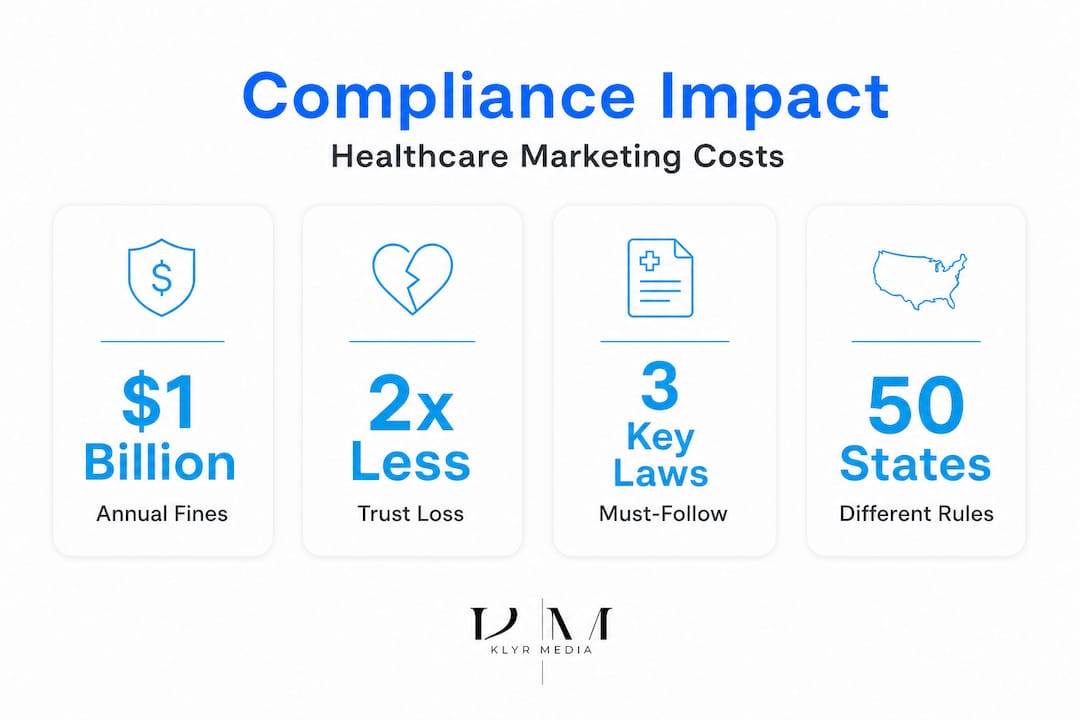
Let's be direct. Non-compliance in healthcare marketing isn't just a paperwork problem. It's a threat to your license, your reputation, and your ability to stay open. For large hospital networks, a fine might be an inconvenience. For an independent pharmacy or a small clinic, it can be devastating.
The risks break down into three categories:
- Legal and financial exposure. Fines, corrective advertising mandates, and litigation costs can pile up fast. The FTC and HHS don't reserve enforcement for big players.
- Loss of patient trust. One misleading ad, one unconsented email blast, and patients walk. In a local market, word travels fast.
- Operational disruption. Investigations freeze marketing programs and pull your team's attention away from patient care.
"FTC Section 5 mandates truthful, non-misleading ads with substantiation by competent scientific evidence for health claims, applying heightened scrutiny to healthcare advertising."
That last phrase, "heightened scrutiny," is important. Healthcare gets held to a higher standard than most industries because the stakes for patients are higher. An exaggerated claim about a shoe sale is annoying. An exaggerated claim about a medication or treatment can cause real harm.
HIPAA adds another layer. Any marketing that uses protected health information (PHI), which includes names, diagnoses, prescription data, or anything that can identify a patient, must follow strict rules around consent and disclosure. Running HIPAA-compliant ads isn't optional. It's the baseline.

For independent pharmacies especially, the FDA's oversight of drug-related claims means you can't casually mention that a product "cures" or "treats" a condition without running into serious regulatory risk. The foundational lesson here: compliance protects you, but it also protects your patients. That alignment is something you can actually market.
Now that we understand the stakes, let's look at which regulations matter most.
Key regulations shaping healthcare marketing
Here's the honest truth about healthcare marketing regulations. There are a lot of them, and they come from multiple directions at once. Federal rules, state rules, professional board rules. Each one has its own scope and its own enforcement teeth.
| Regulation | Who it applies to | What it restricts |
|---|---|---|
| FTC Act (Section 5) | All marketers | False or unsubstantiated claims |
| HIPAA | Any entity using PHI | Unauthorized use of patient data in marketing |
| FDA Rules | Pharma, device marketers | Off-label claims, unfair benefit/risk balance |
| Anti-Kickback Statute (AKS) | Anyone billing federal programs | Incentives designed to induce referrals |
| Stark Law | Physicians and referral sources | Financial relationships that affect referrals |
| State Laws | Varies by state | Disclaimers, opt-in rules, "specialist" title restrictions |
The FDA regulates marketing of drugs and devices, prohibiting off-label claims and requiring what's called "fair balance," meaning your ad must present both benefits and risks proportionally. If you run a campaign promoting a compounded medication or a medical device your clinic uses, you can't cherry-pick only the benefits. The risks need representation. Check the FDA approval status of any product before building a campaign around it.
The Anti-Kickback Statute and Stark Law are where a lot of independent providers get tripped up in their marketing. AKS prohibits any remuneration intended to induce referrals for services covered by Medicare, Medicaid, or other federal programs. "Remuneration" is broad. It includes cash, gifts, free services, or co-marketing arrangements where one party benefits from another's referral stream. Even if you didn't intend it as a kickback, partial intent is enough to trigger a violation.
State-level rules add even more complexity. State laws layer on specifics like mandatory disclaimers on ads, restrictions on using the word "specialist" without board certification, and stricter opt-in requirements for email or SMS marketing. California, Texas, New York, and Florida all have additional rules that go beyond federal minimums. If your clinic or pharmacy operates in multiple states, or if you're running digital ads that could reach patients in different states, you need to understand each state's specific requirements. Staying current on data privacy in healthcare across jurisdictions is a continuous process, not a one-time checkbox.
The financial and operational cost of getting this wrong is significant. Healthcare organizations collectively pay hundreds of millions of dollars annually in regulatory settlements, a burden that falls hardest on smaller providers with fewer legal resources.
With the regulatory landscape defined, let's see how to apply these rules to real marketing activity.
Common compliance pitfalls and how to avoid them
Most violations aren't the result of intentional deception. They happen because marketing teams move fast, compliance review gets skipped, or someone assumes a tactic that works in another industry is fine in healthcare. Here are the five most common traps and how to avoid each one.
-
Unsubstantiated health claims. Saying your pharmacy's supplement "boosts immunity" or your clinic "cures" chronic pain without peer-reviewed evidence to back it up is a direct FTC risk. Fix: require a documented evidence trail for every health claim. If you can't cite a credible source, rephrase or remove the claim.
-
Off-label drug or device promotion. Promoting a medication for a use not listed on its FDA-approved label, even subtly, crosses a serious regulatory line. Fix: audit all campaign copy against the approved labeling for any drug or device you mention, and get a second review from someone who knows the FDA framework.
-
Improper referral incentives. Offering a physician's office free marketing materials, co-branded collateral, or event sponsorships in exchange for referrals can trigger AKS and Stark Law scrutiny. Fix: document the fair market value of every co-marketing arrangement and make sure no benefit is contingent on referral volume.
-
Missing disclosures. Testimonials, before-and-after images, and paid endorsements all require clear disclosure in healthcare advertising. Fix: build disclosure templates into your content workflow so no campaign goes live without the required language.
-
Sloppy opt-ins. Collecting patient emails or phone numbers without proper HIPAA-compliant consent, or sending marketing messages without clear opt-out mechanisms, is a common gap. Fix: review your entire opt-in flow at least twice a year. Verify that your SMS and email platforms are compliant with state and federal requirements.
Pro Tip: Create a standing 30-minute monthly compliance review on your marketing calendar. Pull five random pieces of active content and run them through a simple checklist: Is every health claim substantiated? Are disclosures visible? Is all patient data usage consented? This habit alone catches problems before they become violations.
Consistent auditing also helps with local patient acquisition because compliant marketing tends to be cleaner, more specific, and more credible to local audiences.
Knowing what to avoid, how can compliant marketing actually drive better business results?
How compliance enables smarter marketing and growth
Here's the counterintuitive truth: the practices that make your marketing compliant are the same ones that make it more effective. Let's compare the outcomes directly.
| Marketing approach | Short-term result | Long-term result |
|---|---|---|
| Compliance-blind | Faster campaign launches, bolder claims | Fines, retracted campaigns, damaged reputation |
| Compliance-driven | Slightly longer review cycles | Sustained trust, consistent messaging, lower legal risk |
| Over-cautious avoidance | Very low risk | Almost no marketing, stalled growth |
The systematic approach, using defined policies and regular reviews, opens up creative space instead of closing it. When your team knows exactly what the boundaries are, they stop second-guessing every campaign and start building within a framework that everyone trusts.
Here's what compliant, well-structured healthcare marketing actually delivers:
- Higher patient trust. Patients who see clear, honest, substantiated messaging trust your practice more. That trust converts into appointments and word-of-mouth referrals.
- Reduced rework. Every campaign that gets pulled or revised after launch costs money and time. Compliance review up front eliminates most of that waste.
- Sustainable lead generation. FTC-compliant advertising that makes honest, substantiated claims builds a consistent pipeline without the volatility of risky tactics.
- Stronger referral relationships. Physicians and other providers want to refer patients to practices they trust. A transparent, compliant reputation is a referral magnet.
- Better digital performance. Compliant copy, optimized healthcare websites, and clean data practices tend to align with what search engines reward as well.
Pro Tip: Turn compliance into a visible selling point. Add a simple statement to your website and intake materials: "We follow all HIPAA and FTC guidelines to protect your information and provide honest health information." Patients notice. It differentiates you from practices that say nothing about how they handle data and marketing.
The bigger picture here is that digital healthcare transformation requires a solid compliance foundation to scale. You can't automate patient outreach, run targeted ad campaigns, or build an SEO strategy on shaky regulatory ground. Compliance is the bedrock that makes growth possible. If you're building out your smart marketing keywords strategy, for instance, your keyword choices and landing page claims both need to pass a compliance review before going live.
To pull it all together, let's share insights drawn from what most experts overlook.
Why the best healthcare marketers see compliance as an asset, not a restraint
Here's an uncomfortable observation. The practices that grow fastest in competitive local markets aren't the ones with the most aggressive marketing. They're the ones with the most consistent, trustworthy marketing. And consistency requires compliance.
We've worked with independent pharmacies and clinics that were afraid to market at all because they couldn't tell where the lines were. They weren't non-compliant, they were just paralyzed. That over-avoidance is just as dangerous as ignorance. If you're not showing up in local searches, not running any digital campaigns, and not communicating proactively with patients, you are losing ground to chains and online competitors who are not sitting still.
The teams that figure this out fastest are the ones that treat compliance as a creative brief, not a restriction. When you know you can't make unsubstantiated claims, you invest in getting actual clinical evidence to cite. When you know you can't use patient data without consent, you build a better consent workflow that also doubles as a patient engagement touchpoint. Every constraint pushes you toward a more credible, more patient-centered approach.
Proactive partnership between your marketing team and your compliance officer is the difference maker. Most compliance officers don't want to block campaigns. They want to avoid violations. When marketers bring them into the process early, instead of submitting final copy for review at the last second, you get faster approvals and better campaigns. The adversarial dynamic between marketing and compliance is almost always a process problem, not a people problem.
The smartest move you can make for SEO for healthcare growth is to build your content strategy around compliant, well-substantiated information that genuinely answers patient questions. That approach earns search rankings, builds trust, and keeps you safe from regulatory risk all at once.
Compliance isn't red tape. It's the framework that lets you market confidently, consistently, and at scale.
Get hands-on help with compliant healthcare marketing
Running a compliant marketing program while also managing patient care, staff, and operations is a lot to ask of any independent practice. You don't have to figure it out alone.
At KLYR Media, we build marketing systems designed specifically for independent pharmacies and medical clinics, built around HIPAA-compliant web design, healthcare SEO solutions, and automated patient engagement workflows that respect regulatory requirements from the ground up. Every strategy we develop is tested against the compliance framework your practice operates in. If you're ready to grow without the guesswork, explore our digital healthcare transformation solutions and request a free audit of your current marketing setup. Let's build something that grows your practice and keeps it protected.
Frequently asked questions
What happens if my healthcare marketing doesn't meet FTC standards?
You risk fines, mandated corrective advertising, and potential legal action, especially if your campaigns include unsubstantiated health claims that mislead patients about a treatment's effectiveness.
Are off-label drug promotions allowed in healthcare marketing?
No. FDA regulations prohibit off-label promotion in any marketing material, and your campaigns must present a fair balance of benefits and risks for any drug or device you mention.
How can I ensure my healthcare marketing is compliant across different states?
Audit each state's specific requirements for disclaimers, opt-ins, and professional title restrictions, because state laws vary significantly across California, Texas, New York, Florida, and elsewhere, and consult local legal counsel for market-specific details.
What types of incentives are not allowed under the Anti-Kickback Statute?
Any direct or indirect payment intended to generate referrals for federally covered services can violate AKS, and even partial intent to reward referrals is enough to trigger an enforcement action.
Recommended
- How to Optimize Google Ads for Healthcare: HIPAA-Compliant Patient Acquisition | Blog
- How to Promote Your Healthcare Clinic Location: Local Patient Acquisition Strategies | Blog
- Medical Clinic Solutions | Custom Practice Management Software | KLYR Media | KLYR Media
- Healthcare SEO Agency | Pharmacy SEO & Medical Practice SEO | KLYR Media
